Current issue
Online first
Archive
About the Journal
Editorial Office
Editorial Board
Copy right and self-archiving policy
Peer review process
Instructions for Reviewers
Printed version subscription
Abstracting and indexing
Contact
Instructions for Authors
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
Article publication charges
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
ORIGINAL PAPER
Chestnut tannin supplementation can improve immune response and kidney function in prepartum dairy cows
1
University of Belgrade, Faculty of Veterinary Medicine, Department of Ruminant and Swine Diseases,
Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
2
University of Belgrade, Faculty of Veterinary Medicine, Department of Physiology and Biochemistry, Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
3
University of Belgrade, Faculty of Veterinary Medicine, Department of Reproduction, Fertility and Artificial Insemination, Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
4
University of Belgrade, Faculty of Veterinary Medicine, Department of General Education, Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
Publication date: 2023-11-17
Corresponding author
D. Kirovski
University of Belgrade, Faculty of Veterinary Medicine, Department of Physiology and Biochemistry, Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
University of Belgrade, Faculty of Veterinary Medicine, Department of Physiology and Biochemistry, Bulevar Oslobodjenja 18, 11000 Belgrade, Serbia
J. Anim. Feed Sci. 2024;33(2):185-192
KEYWORDS
TOPICS
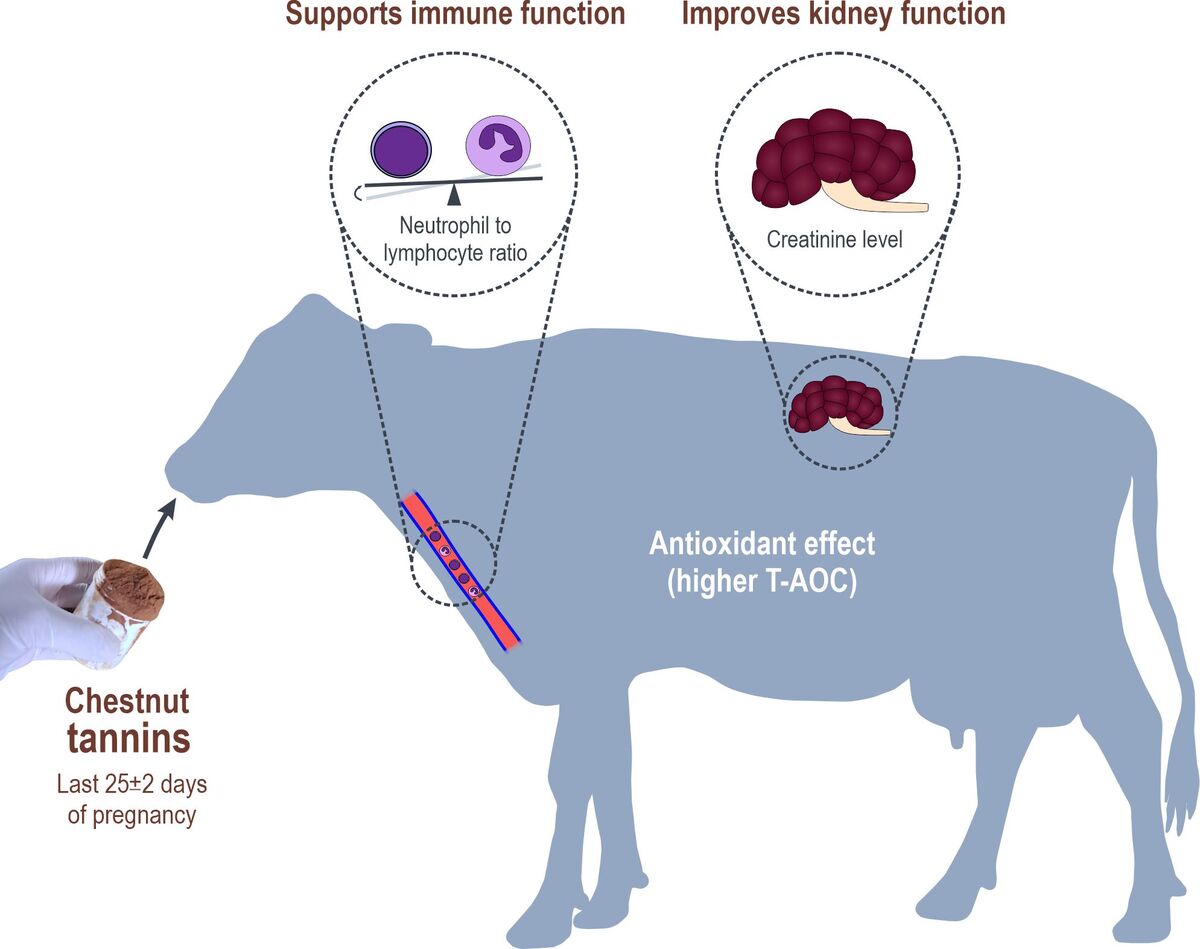
ABSTRACT
Due to their antiketogenic and antioxidant effects, chestnut
tannins may offer a viable approach to manage the impaired immune and
renal functions in transition cows. This study aimed to investigate the effects of
dietary supplementation with chestnut tannins on haematological, biochemical
and antioxidant indices, as well as cortisol levels in prepartum dairy cows.
Forty multiparous Holstein cows were divided into two homogeneous groups
(n = 20): a control (CON), and an experimental group (CNT) receiving
20 g/day of chestnut tannins for the last 25 ± 2 days of pregnancy. Haematological
and biochemical indices, cortisol concentration and total antioxidant capacity
(T-AOC) were measured in blood samples collected 25 (day −25) and 5 days
(day −5) before the expected parturition. The addition of chestnut tannins exerted
no significant effect on red blood cells indices; however, white blood cell (P =
0.02), lymphocyte (P = 0.05) and platelet (P < 0.01) counts were higher, while the
neutrophil to lymphocyte ratio (P = 0.03) was lower on day −5 in the CNT group
compared to the CON group. Significantly higher values of T-AOC (P = 0.03)
and significantly lower levels of triglycerides (P = 0.03) and gamma-glutamyl
transferase (P = 0.02) were also found in CNT compared to CON on day −5. The
improved haematological profile in CNT cows was accompanied by lower serum
creatinine concentration (P = 0.04), while total protein, calcium, phosphorus and
cortisol did not differ significantly between CNT and CON cows. These data
demonstrate that dietary chestnut tannin supplementation in a close-up diet has
antioxidant and anti-inflammatory effects, and could potentially mitigate immune
suppression and kidney dysfunction near parturition. Further research should be
conducted concerning the mechanisms underlying these responses.
FUNDING
This work was supported by the Science
Fund of the Republic of Serbia, under the project:
‘Mitigation of methane production from dairy cattle
farm by nutritive modulation of cow’s metabolism –
MitiMetCattle’ No 7750295.
CONFLICT OF INTEREST
The Authors declare that there is no conflict of
interest.
REFERENCES (35)
1.
Ahmad S.T., Sultana S., 2012. Tannic acid mitigates cisplatin-induced nephrotoxicity in mice. Hum. Exp. Toxicol. 31, 145–156, https://doi.org/10.1177/096032....
2.
Bešlo D., Došlić G., Agić D., Rastija V., Šperanda M., Gantner V., Lučić B., 2022. Polyphenols in ruminant nutrition and their effects on reproduction. Antioxidants. 11, 970. https://doi:10.3390/antiox1105....
3.
Bionaz M.E.A., Trevisi E., Calamari L.U.I.G.I., Librandi F., Ferrari A., Bertoni G., 2007. Plasma paraoxonase, health, inflammatory conditions, and liver function in transition dairy cows. J. Dairy Sci. 90, 1740–1750, https://doi.org/10.3168/jds.20....
4.
Brscic M., Cozzi G., Lora I., Stefani A.L., Contiero B., Ravarotto L., Gottardo F., 2015. Reference limits for blood analytes in Holstein late-pregnant heifers and dry cows: Effects of parity, days relative to calving, and season. J. Dairy Sci. 98, 7886–7892, https://doi.org/10.3168/jds.20....
5.
Dorshkind K., Horseman N.D., 2000. The roles of prolactin, growth hormone, insulin-like growth factor-I, and thyroid hormones in lymphocyte development and function: insights from genetic models of hormone and hormone receptor deficiency. Endocr. Rev. 21, 292–312, https://doi.org/10.1210/edrv.2....
6.
Drackley J.K., Dann H.M., Douglas N., Guretzky N.A.J., Litherland N.B., Underwood J.P., Loor J.J., 2005. Physiological and pathological adaptations in dairy cows that may increase susceptibility to periparturient diseases and disorders. Ital. J. Anim. Sci. 4, 323–344, https://doi.org/10.4081/ijas.2....
7.
Guan R.W., Wang D.M., Wang B.B., Jiang L.Y., Liu J.X., 2020. Prognostic potential of pre-partum blood biochemical and immune variables for postpartum mastitis risk in dairy cows. BMC Vet. Res. 16, 1–11, https://doi.org/10.1186/s12917....
8.
Hong H., Lee E., Lee I.H., Lee S.R., 2019. Effects of transport stress on physiological responses and milk production in lactating dairy cows. Asian-Australas. J. Anim. Sci. 32, 442–451, https://doi.org/10.5713/ajas.1....
9.
Huang Q., Xiuli L., Guoqi Z., Tianming H.U., Yuxi W., 2018. Potential and challenges of tannins as an alternative to in-feed antibiotics for farm animal production. Anim. Nutr. 4, 137–150, https://doi.org/10.1016/j.anin....
10.
Issi M., Gül Y., Başbuğ O., 2016. Evaluation of renal and hepatic functions in cattle with subclinical and clinical ketosis. Turk. J. Vet. Anim. Sci. 40, 47–52, http://dx.doi.org/10.3906/vet-....
11.
Jayanegara A., Goel G., Makkar H.P., Becker K., 2015. Divergence between purified hydrolysable and condensed tannin effects on methane emission, rumen fermentation and microbial population in vitro. Anim. Feed Sci. Tech. 209, 60–68, https://doi.org/10.1016/j.anif....
12.
Jones M.L., Allison R.W., 2007. Evaluation of the ruminant complete blood cell count. Vet. Clin. North Am. Food Anim. Pract. 23, 377–402, https://doi.org/10.1016/j.cvfa....
13.
Jonsson N.N., Fortes M.R.S., Piper E.K., Vankan D.M., de Cisneros J.P.J., Wittek T., 2013. Comparison of metabolic, hematological, and peripheral blood leukocyte cytokine profiles of dairy cows and heifers during the periparturient period. J. Dairy Sci. 96, 2283–2292, https://doi.org/10.3168/jds.20....
14.
Knegsel A.V., Hammon H.M., Bernabucci U. et al., 2014. Metabolic adaptation during early lactation: key to cow health, longevity and a sustainable dairy production chain. CABI Rev. 9, 1–15, https://doi.org/10.1079/PAVSNN....
15.
Koenig G., Seneff S., 2015. Gamma-glutamyltransferase: a predictive biomarker of cellular antioxidant inadequacy and disease risk. Dis. Markers 2015, 818570, https://doi.org/10.1155/2015/8....
16.
Lacetera N., Scalia D., Bernabucci U., Ronchi B., Pirazzi D., Nardone A., 2005. Lymphocyte functions in overconditioned cows around parturition. J. Dairy Sci. 88, 2010–2016, https://doi.org/10.3168/jds.s0....
17.
Liu H.W., Zhou D.W., Li K., 2013. Effects of chestnut tannins on performance and antioxidative status of transition dairy cows. J. Dairy Sci. 96, 5901–5907, https://doi.org/10.3168/jds.20....
18.
Liu J.C., Hsu F.L., Tsai J.C., Chan P., Liu J.Y., Thomas G.N., Tomlinson B., Lo M.Y., Lin J.Y., 2003. Antihypertensive effects of tannins isolated from traditional Chinese herbs as non-specific inhibitors of angiontensin converting enzyme. Life Sci. 73, 1543–1555, https://doi.org/10.1016/s0024-....
19.
Ma S., Zhu X.Y., Eirin A., Woollard J.R., Jordan K.L., Tang H., Lerman A., Lerman L. O., 2016. Perirenal fat promotes renal arterial endothelial dysfunction in obese swine through tumor necrosis factor-α. J. Urol. 195, 1152–1159, https://doi.org/10.1016/j.juro....
20.
NRC (National Research Council), 2001. Nutrient requirements of dairy cattle, 7th Revised Ed., Washington, DC.
21.
Olafadehan O.A., Adewumi M.K., Okunade S.A., 2014. Effects of feeding tannin-containing forage in varying proportion with concentrate on the voluntary intake, haematological and biochemical indices of goats. Trakia J. Sci. 12, 73–81.
22.
Orzuna-Orzuna J.F., Dorantes-Iturbide G., Lara-Bueno A., Mendoza-Martínez G.D., Miranda-Romero L.A., Hernández-García P.A., 2021. Effects of dietary tannins’ supplementation on growth performance, rumen fermentation, and enteric methane emissions in beef cattle: a meta-analysis. Sustainability 13, 7410. https://doi.org/10.3390/su1313....
23.
Piazza S., Fumagalli M., Martinelli G., Pozzoli C., Maranta N., Angarano M., Sangiovanni E., Dell’Agli M., 2022. Hydrolyzable tannins in the management of Th1, Th2 and Th17 inflammatory-related diseases. Molecules 27, 7593, https://doi.org/10.3390/molecu....
24.
Prodanović R., Korićanac G., Vujanac I., Djordjević A., Pantelić M., Romić S., Stanimirović Z., Kirovski D., 2016. Obesity-driven prepartal hepatic lipid accumulation in dairy cows is associated with increased CD36 and SREBP-1 expression. Res. Vet. Sci. 107, 16–19, https://doi.org/10.1016/j.rvsc....
25.
Prodanović R., Nedić S., Simeunović P., Borozan S., Nedić S., Bojkovski J., Kirovski D., Vujanac I., 2021. Effects of chestnut tannins supplementation of prepartum moderate yielding dairy cows on metabolic health, antioxidant and colostrum indices. Ann. Anim. Sci. 21, 609–621, https://doi.org/10.2478/aoas-2....
26.
Prodanović R., Nedić S., Vujanac I., Bojkovski J., Nedić S., Jovanović L., Kirovski D., Borozan S., 2023. dietary supplementation of chestnut tannins in prepartum dairy cows improves antioxidant defense mechanisms interacting with thyroid status. Metabolites 13, 334, https://doi.org/10.3390/metabo....
27.
Qi J., Gan L., Fang J., et al., 2022. Beta-hydroxybutyrate: a dual function molecular and immunological barrier function regulator. Front. Immunol. 13, 805881, https://doi.org/10.3389/fimmu.....
28.
Re R., Pellegrini N., Proteggente A., Pannala A., Yang M., Rice-Evans C., 1999. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 26, 1231–1237, https://doi.org/10.1016/s0891-....
29.
Santillo A., Ciliberti M.G., Ciampi F., Luciano G., Natalello A., Menci R., Caccamo M., Sevi A., Albenzio M., 2022. Feeding tannins to dairy cows in different seasons improves the oxidative status of blood plasma and the antioxidant capacity of cheese. J. Dairy Sci. 105, 8609–8620, https://doi.org/10.3168/jds.20....
30.
Steenge G.R., Lambourne J., Casey A., Macdonald I.A., Greenhaff P.L., 1998. Stimulatory effect of insulin on creatine accumulation in human skeletal muscle. Am. J. Physiol. 275, 974–979, https://doi.org/10.1152/ajpend....
31.
van Drongelen J., de Vries R., Lotgering F.K., Smits P., Spaanderman M.E., 2014. Functional vascular changes of the kidney during pregnancy in animals: A systematic review and meta-analysis. PLoS One 9, e112084, https://doi.org/10.1371/journa....
32.
Waqas M., Salman M., Sharif M.S., 2023. Application of polyphenolic compounds in animal nutrition and their promising effects. J. Anim. Feed Sci. 32, 233–256, https://doi.org/10.22358/jafs/....
33.
Wankhade P.R., Manimaran A., Kumaresan A., Jeyakumar S., Ramesha K.P., Sejian V., Rajendran D., Varghese, M.R., 2017. Metabolic and immunological changes in transition dairy cows: A review. Vet World 10, 1367–1377, https://doi.org/10.14202/vetwo....
34.
Wood D., Quiroz-Rocha G.F., 2010. Normal hematology of cattle. In: D. J. Weiss, K. J. Wardrop. Schalm’s veterinary hematology. Wiley. Ames, IA, 6th Ed., pp. 829–835.
35.
Xiong Y.A., Yu Q.N., Zou J.B., He Y.H., Zhang S.J., Xu R.C., Yang M., 2014. Protective effects of tannins in Sanguisorbae Radix on myelosuppression mice. Chin. Herb. Med. 6, 222–227, https://doi.org/10.1016/S1674-....
CITATIONS (1):
1.
Impact of garlic polysulfide supplementation on biochemical and hematological parameters in dairy cows
Sreten Nedic, Mirjana Gajic, Dusan Bosnjakovic, Ivan Vujanac, Radisa Prodanovic, Jovan Bojkovski, Aleksandra Mitrovic, Milan Milenkovic, Dragan Manojlovic, Ljubomir Jovanovic, Danijela Kirovski, Sveta Arsic
Veterinarski glasnik
Sreten Nedic, Mirjana Gajic, Dusan Bosnjakovic, Ivan Vujanac, Radisa Prodanovic, Jovan Bojkovski, Aleksandra Mitrovic, Milan Milenkovic, Dragan Manojlovic, Ljubomir Jovanovic, Danijela Kirovski, Sveta Arsic
Veterinarski glasnik
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.