Current issue
Online first
Archive
About the Journal
Editorial Office
Editorial Board
Copy right and self-archiving policy
Peer review process
Instructions for Reviewers
Printed version subscription
Abstracting and indexing
Contact
Instructions for Authors
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
Article publication charges
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
ORIGINAL PAPER
Sodium selenite addition promotes the proliferation of bovine
mammary epithelial cells through the Akt-mTOR
signalling pathway
1
Shanxi Agrcultural University, College of Animal Science, Department of Animal Nutrition and Feed Science,
Taigu, 030801, Shanxi, P. R. China
Publication date: 2023-04-25
J. Anim. Feed Sci. 2023;32(3):257-266
KEYWORDS
TOPICS
ABSTRACT
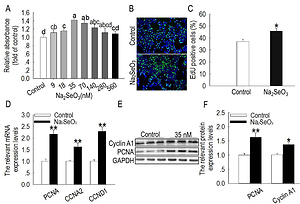
The experiment was designed to evaluate the effect of sodium
selenite (Na2SeO3) on the proliferation of bovine mammary gland epithelial
cells (BMECs), and to assess the underlying mechanisms. The addition of
Na2SeO3 at doses ranging from 18 to 140 nM stimulated BMEC proliferation.
Nevertheless, high doses of Na2SeO3 (> 70 nM) did not further stimulate
BMEC proliferation compared to 35 nM Na2SeO3. Therefore, the addition of
35 nM Na2SeO3 increased the proportion of epithelial cells undergoing DNA
replication, mRNA expression of proliferating cell nuclear antigen (PCNA),
Cyclin A2 (CCNA2) and Cyclin D1 (CCND1), as well as protein expression of
PCNA and Cyclin A1. Transcription and translation of the B-cell lymphoma 2
(BCL2) gene and the ratio of BCL2 to BCL2-associated X 4 (BAX4) genes were
significantly elevated, while mRNA and protein expressions of BAX, caspase 3
(CASP3) and caspase 9 (CASP9) genes were significantly reduced as a result of
35 nM Na2SeO3 supplementation. Additionally, both the Akt and mTOR signalling
pathways were activated by 35 nM Na2SeO3. On the other hand, the stimulation
of BMEC proliferation, altered expression of proliferative and apoptotic genes
and proteins, as well as mTOR signalling pathway activation caused by 35 nM
Na2SeO3 addition was suppressed by the Akt inhibitor (AKT-IN-1). Likewise,
rapamycin-mediated suppression of mTOR completely reversed the 35 nM
Na2SeO3-stimulated BMEC proliferation and alteration of proliferous gene and
protein expressions, without affecting mRNA or protein expression of genes
related to apoptosis and the Na2SeO3-activated Akt signalling pathway. In
conclusion, the results implicated that the proliferation of BMECs was stimulated
by 35 nM Na2SeO3 through the Akt-mTOR signalling pathway.
FUNDING
This work was supported by the grant from the
Science and Technology Innovation Fund Project
of Shanxi Agricultural University (Grant No.
2018YJ36) and the Key Research and Development
project of Shanxi Province (201903D221012).
CONFLICT OF INTEREST
The Authors declare that there is no conflict of
interest.
REFERENCES (32)
1.
Akers R.M. (Editor), 2002. Lactation and the mammary gland. 1st Edition. Wiley-Blackwell. Hoboken, NJ (USA)
2.
Akers R.M., 2017. A 100-Year Review: mammary development and lactation. J. Dairy Sci. 100, 10332–10352, http://dx.doi.org/10.3168/jds....
3.
Bae D., Chon J.W., Kim D.H., Kim H., Seo K.H., 2020. Effect of folic acid supplementation on proliferation and apoptosis in bovine mammary epithelial (MAC-T) cells. Anim. Biotechnol. 33, 13–21, http://dx.doi.org/10.1080/1049...
4.
Bertoli C., Skotheim J.M., de Bruin R.A.M., 2013. Control of cell cycle transcription during G1 and S phases. Nat. Rev. Mol. Cell Biol. 14, 518–528, https://doi.org/10.1038/nrm362...
5.
De Cicco P., Panza E., Ercolano G., Armogida C., Sessa G., Pirozzi G., Cirino G., Wallace J.L., Ianaro A., 2016. ATB-346, a novel hydrogen sulfide-releasing anti-inflammatory drug, induces apoptosis of human melanoma cells and inhibits melanoma development in vivo. Pharmacol. Res. 114, 67–73, http://dx.doi.org/10.1016/j.ph...
6.
He S.L., Guo X., Tan W.H., Su X.H., Li J.P., Pan W., Qiu H.Y., 2016. Effect of selenium deficiency on phosphorylation of the AMPK pathway in rats. Biol. Trace Elem. Res. 169, 254–260, http://dx.doi.org/10.1007/s120...
7.
Huang J., Qin Y.Y., Lin C.F., Huang X.G., Zhang F.R., 2021. MTHFD2 facilitates breast cancer cell proliferation via the AKT signaling pathway. Exp. Ther. Med. 22, 703, http://dx.doi.org/10.3892/etm....
8.
Huang Z., Rose A.H., Hofmann P.R., 2012. The role of selenium in infammation and immunity: from molecular mechanisms to therapeutic opportunities. Antioxid. Redox. Signal. 16, 705–743, http://dx.doi.org/10.1089/ars....
9.
Jiang D., Cho W.C., Li Z., Xu X., Qu Y., Jiang Z., Guo L., Xu, G., 2017. MiR-758-3p suppresses proliferation, migration and invasion of hepatocellular carcinoma cells via targeting MDM2 and mTOR. Biomed. Pharmacother. 96, 535–544, http://dx.doi.org/10.1016/j.bi...
10.
Kim S.W., Wu G., 2009. Regulatory role for amino acids in mammary gland growth and milk synthesis. Amino Acids 37, 89–95, http://dx.doi.org/10.1007/s007...
11.
Li F., Lutz P.B., Pepelyayeva Y., Arnér E.S.J., Bayse C.A., Rozovsky S., 2014. Redox active motifs in selenoproteins. Proc. Natl. Acad. Sci. U. S. A. 111, 6976–6981, http://dx.doi.org/10.1073/pnas...
12.
Li L., Liu L., Qu B., Li X., Gao X., Zhang M., 2017. Twinfilin 1 enhances milk bio-synthesis and proliferation of bovine mammary epithelial cells via the mTOR signaling pathway. Biochem. Biophys. Res. Commun. 492, 289–294, http://dx.doi.org/10.1016/j.bb...
13.
Macrina A.L., Kauf A.C.W., Kensinger R.S., 2011. Effect of bovine somatotropin administration during induction of lactation in 15-month-old heifers on production and health. J. Dairy Sci. 94, 4566–4573, http://dx.doi.org/10.3168/jds....
14.
McCabe C.J., Suarez-Trujillo A., Teeple K.A., Casey T.M., Boerman J.P., 2021. Chronic prepartum light-dark phase shifts in cattle disrupt circadian clocks, decrease insulin sensitivity and mammary development, and are associated with lower milk yield through 60 days postpartum. J. Dairy Sci. 104, 2422–2437, http://dx.doi.org/10.3168/jds....
15.
Meng Y., Zhang J., Zhang F. et al., 2017. Lauric acid stimulates mammary gland development of pubertal mice through activation of GPR84 and PI3K/Akt signaling pathway. J. Agric. Food Chem. 65, 95–103, http://dx.doi.org/10.1021/acs....
16.
Molenaar A.J., Maclean P.H., Gilmour M.L., Draganova I.G., Symes C.W., Margerison J.K., Mcmahon C.D., 2020. Effect of whole-milk allowance on liveweight gain and growth of parenchyma and fat pads in the mammary glands of dairy heifers at weaning. J. Dairy Sci. 103, 5061–5069, http://dx.doi.org/10.3168/jds....
17.
Najafnejad B., Aliarabi H., Tabatabaei M.M., Alimohamady R., 2013. Effects of different forms of selenium supplementation on production performance and nutrient digestibility in lactating dairy cattle. In: Proceedings of the Second International Conference on Agriculture and Natural Resources. 25–26 December 2013. Kermanshah (Iran).
18.
Pan J., Li K., Huang W., Zhang X., 2017. MiR-137 inhibited cell proliferation and migration of vascular smooth muscle cells via targeting IGFBP-5 and modulating the mTOR/STAT3 signaling. PLoS One 12, e0186245, http://dx.doi.org/10.1371/jour...
19.
Park S.Y., Mi S.J., Chang W.H., Yu H.S., Jang S.B., 2016. Structural and functional insight into proliferating cell nuclear antigen. J. Microbiol. Biotechnol. 26, 637–647, http://dx.doi.org/10.4014/jmb....
20.
Pisani C., Ramella M., Boldorini R., Loi G., Billia M., Boccafoschi F., Volpe A., Krengli M., 2020. Apoptotic and predictive factors by Bax, caspases 3/9, Bcl-2, p53 and Ki-67 in prostate cancer after 12 Gy single-dose. Sci. Rep. 10, 7050, http://dx.doi.org/10.1038/s415...
21.
SAS Institute, 2002. SAS/STAT®9.0 User's Guide. SAS Institute Inc. Cary, NC (USA)
22.
Shamas-Din A., Kale J., Leber B., Andrews D.W., 2013. Mechanisms of action of Bcl-2 family proteins. Cold Spring Harbor Perspect. Biol. 5, a008714, http://dx.doi.org/10.1101/cshp...
23.
Shi L., Duan Y., Yao X., Song R., Ren Y., 2020. Effects of selenium on the proliferation and apoptosis of sheep spermatogonial stem cells in vitro. Anim. Reprod. Sci. 215, 106330, https://doi.org/10.1016/j.anir...
24.
Souza F.N., Blagitz M.G., Latorre A.O., Mori C.S., Sucupira M.C.A., Della Libera A.M.M.P., 2012. Effects of in vitro selenium supplementation on blood and milk neutrophils from dairy cows. Pesq. Vet. Bras. 32, 174–178, https://doi.org/10.1590/S0100-...
25.
Ullah H., Khan R.U., Mobashar M., Ahmad S., Sajid A., Khan N.U., Usman T., Khattak I., Khan H., 2019. Effect of yeast-based selenium on blood progesterone, metabolites and milk yield in Achai dairy cows. Ital. J. Anim. Sci. 18, 1445–1450, https://doi.org/10.1080/182805...
26.
Wang C., Liu Q., Yang W.Z., Dong Q., Yang X.M., He D.C., Zhang P., Dong K.H., Huang Y.X., 2009. Effects of selenium yeast on rumen fermentation, lactation performance and feed digestibilities in lactating dairy cows. Livest. Sci. 126, 239–244, https://doi.org/10.1016/j.livs...
27.
Xiao J., Khan M.Z., Ma Y., Alugongo G.M., Ma J., Chen T., Khan A., Cao Z., 2021. The antioxidant properties of selenium and vitamin E; their role in periparturient dairy cattle health regulation. Antioxidants 10, 1555, https://doi.org/10.3390/antiox...
28.
Zhang B.Q., Guo Y.M., Yan S.M., Guo X.Y., Zhao Y.L., Shi B.L., 2020. The protective effect of selenium on the lipopolysaccharide-induced oxidative stress and depressed gene expression related to milk protein synthesis in bovine mammary epithelial cells. Biol. Trace Elem. Res. 197, 141–148, https://doi.org/10.1007/s12011...
29.
Zhang J., Ye J., Yuan C., Fu Q., Zhang F., Zhu X., Wang L., Gao P., Shu G., Jiang Q., Wang S., 2018. Exogenous H2S exerts biphasic effects on porcine mammary epithelial cells proliferation through PI3K/Akt-mTOR signaling pathway. J. Cell. Physiol. 233, 7071–7081, http://dx.doi.org/10.1002/jcp....
30.
Zhang Y., Xu Y., Chen B., Zhao B., Gao X.J., 2021. Selenium deficiency promotes oxidative stress-induced mastitis via activating the NF-κB and MAPK pathways in dairy cow. Biol. Trace Elem. Res. 200, 2716–2726, http://dx.doi.org/10.1007/s120...
31.
Zhao K., Liu H.Y., Zhou M.M., Liu J.X., 2010. Establishment and characterization of a lactating bovine mammary epithelial cell model for the study of milk synthesis. Cell Biol. Int. 34, 717–721, http://dx.doi.org/10.1042/CBI2...
32.
Zhong J.X., Jin S.S., Wu K.S., Yu G.C., Tu L.L., Liu L., 2022. Effect of nano-selenium loaded with lycium barbarum polysaccharide on the proliferation of lens epithelial cells after UVB damage in vitro. Int. J. Ophthalmol. 15, 9–14, http://dx.doi.org/10.18240/ijo...
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.